
Dr Megan Shelden
Senior Lecturer
School of Agriculture, Food and Wine
College of Science
Eligible to supervise Masters and PhD - email supervisor to discuss availability.
Megan is a Senior Lecturer and Mortlock Fellow at the Waite Campus, Adelaide University. She has extensive knowledge and expertise in plant biology. She is passionate about enhancing food security by improving crop yields in challenging soils, such as those affected by salinity and drought.Salinity affects an estimated 5.7 million hectares of land in Australia, posing significant environmental and agricultural challenges. The area of land affected by salinity will likely be accelerated with climate change. The economic cost associated with saline soils to the Australian farming industry is estimated to be around $1.5 billion annually. Megan's lab group is focused on unravelling the secrets of plants below the ground. Plant roots have a remarkable ability to adapt to stressful conditions like salinity, while continuing to grow. She explores how plant roots respond to these environmental stressors by altering their growth to increase their ability to access nutrients and water in the soil. Her expertise is multi-faceted and spans various disciplines, including root biology, plant physiology, plant biochemistry, molecular biology, and functional genomics.Currently, Megan is working on uncovering the molecular mechanisms that control and regulate root growth in response to salinity using barley as a model cereal crop. By studying barley, she aims to gain valuable information that can be applied to increase salinity tolerance in other vital Australian cereal crops, like wheat, to help reduce the significant financial cost of salinity on grain yield. Megan's expertise in her field extends beyond her research. She actively contributes to the Australian Society of Plant Scientists (ASPS), having served on the committee as a discipline representative and contributed to the establishment of a Gender, Equity and Diversity policy. Her involvement in the society allows her to connect with fellow experts and stay at the forefront of scientific advancements. Passionate about inspiring the next generation of plant and agricultural scientists through her teaching at the University of Adelaide, Megan encourages students to explore the fascinating world of plant science. She firmly believes in the power of education and hopes to nurture a diverse and inclusive community of budding scientists. Megan engages in promoting STEM (Science, Technology, Engineering, and Mathematics) to the wider community through events such as National Science Week. Her contribution to National Science Week was recognised by being the recipient of the ASPS Education and Outreach Award in 2022. She is a strong advocate for Women in STEM and has initiated the Women in Crop Science network at the Waite campus. Megan's innovative approaches to understanding crop root growth in challenging soils and ensuring food security for future generations, has secured over $1.6 million in grant funding from nationally competitive grant programs. She also has several highly cited publications in journals such as The Plant Journal and Journal of Experimental Botany. Megan is a committed researcher and passionate educator in plant science. Her work in understanding root systems and improving crop yields in challenging soils aims to make a significant contribution to agriculture and to a more sustainable future.
Current Projects:
Exploiting root traits for improved yields in salty soils
Megan's research is aimed at understanding the mechanisms that allow crop root systems to adapt to salinity and drought, thereby maintaining root growth and increasing soil exploration for nutrient and water uptake. Her current research aims to identify the molecular mechanisms that control and regulate root adaptation in response to salinity using barley (Hordeum vulgare L.) as a cereal model. This research will lead to the identification of root traits that can be incorporated into breeding programs to develop higher-yielding salt-tolerant crops. For more information see our recent publication in Frontiers in Plant Science:
https://www.frontiersin.org/articles/10.3389/fpls.2023.1120583/full
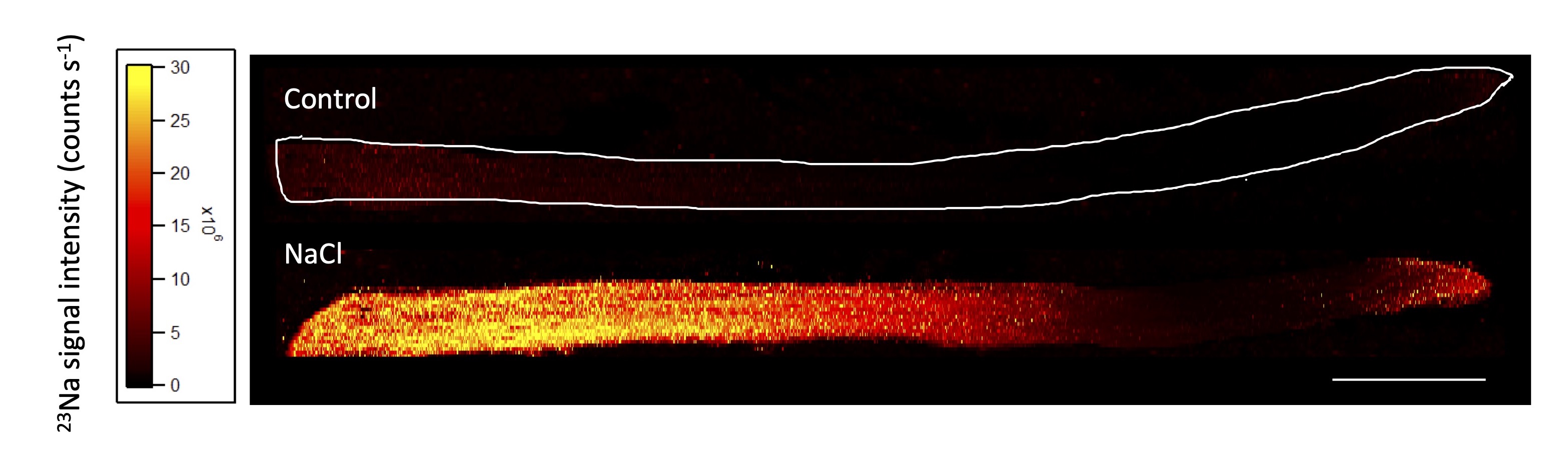
Salt-tolerance in Grapevines
Megan is using grapevine as a model system to understand salt and water movement through plant systems; from the root to shoot. She aims to answer the question of how salt moves across the different tissues and major organs, how salt accumulates in root, leaf, and shoot cells, and how movement and accumulation is controlled by the diversity of transport mechanisms operating in plants. This will be incorporated into biophysical models (in collaboration with UniSA), to quantify the movement of salt through plant organs, tissues, and cells, from root to leaf.
Finding the missing links in salt and water transport in plants
Grain crops and horticultural plants use proteins called aquaporins to move water across cell membranes, however a group of these proteins can also transport some important nutrient ions as well as toxic sodium ions. This project aims to reveal the molecular pathways that regulate water and ion transport via aquaporins using advanced techniques in biophysics and molecular biology. The results will provide novel insights into how plants coordinate and adapt to changing water and salt conditions, addressing a missing link in how ions and water move in and out of plant vacuoles. Benefits include an expanded, innovative range of targets for plant breeding programs to improve plant productivity in our changing climate.
Lab Group
Staff
Dr Sara Jalali
Ms. Daisy Edwards (Research Assistant)
Previous Staff
Dr. Yue Wu (Postdoctoral Fellow, now at PSBA)
Current Students
Ms Zhale Hekmati
Ms Iza Fatima
Mr Caoyang Wang
Completed Students
Dr. Ying Meng
Dr. Apriadi Situmorang (Post-doc at AU)
Research in the news:
Radio interview with ABC rural:
https://www.abc.net.au/news/rural/2015-12-11/sheldon-saline-wheat/7018212
I am recruiting Honours, Masters, and PhD students to work on aspects of the various projects listed above. If interested, please contact Megan directly with an expression of interest and CV (megan.shelden@adelaide.edu.au).
| Date | Position | Institution name |
|---|---|---|
| 2022 - ongoing | Senior Lecturer/ Mortock Fellow | University of Adelaide |
| 2020 - 2021 | Lecturer | The University of Adelaide |
| 2019 - 2020 | Research Fellow | The University of Adelaide |
| 2014 - 2019 | ARC DECRA Fellow | University of Adelaide |
| 2009 - 2013 | Research Fellow | University of Melbourne |
| 2007 - 2009 | Postdoctoral Fellow | Australian National University |
| Date | Institution name | Country | Title |
|---|---|---|---|
| 2023 | Deakin University | Australia | Graduate Certificate in Higher Education (Learning and Teaching) |
| 2008 | University of Adelaide | Australia | PhD |
| 1994 - 1997 | University of Adelaide | Australia | Bachelor of Science (Honours) |
| Year | Citation |
|---|---|
| 2019 | Tyerman, S., Munns, R., Fricke, W., Arsova, B., Barkla, B., Bose, J., . . . Wen, Z. (2019). Energy costs of salinity tolerance in crop plants. In New Phytologist Vol. 221 (pp. 25-29). UK: Wiley. DOI Scopus58 WoS51 Europe PMC37 |
| 2002 | Loughlin, P., Shelden, M. C., Tierney, M. L., & Howitt, S. M. (2002). Structure and function of a model member of the SulP transporter family. In Cell Biochemistry and Biophysics Vol. 36 (pp. 183-190). AUSTRALIA, COOLANGATTA: HUMANA PRESS INC. DOI Scopus14 WoS13 Europe PMC13 |
Grains Research and Development Corporation (2025-2026); Establishing a new paradigm for salinity tolerance in cereals, $222,760
Charlick Endowment Fund (2025); University of Adelaide, $14,700
Research Small Equipment Support Scheme (2024), University of Adelaide, $11,000
UoA-UoN Seed Partnering Grant (2024), University of Adelaide, $10,000
Barbara Kidman Women's Fellowship (2024), University of Adelaide, $30,000
ARC Discovery Project (2022 - 2025); Finding the missing links in salt and water transport in plants, $489,236
ARC Discovery Project (2020 - 2023); Root-to-shoot: modeling the salt stress response of a plant vascular system, $420,000
ARC DECRA Fellow (2014 - 2017); Getting to the root of salt-tolerance in the model cereal crop, barley, $394,570
PRIF Catalyst Research Grant (2015 - 2016); Screening for salt-tolerance in wheat using impedance spectroscopy: A novel technique to reveal performance of the hidden half, $30,000
I contribute to teaching in:
Foundations in Plant Science II
Viticulture II /A
| Date | Role | Research Topic | Program | Degree Type | Student Load | Student Name |
|---|---|---|---|---|---|---|
| 2025 | Co-Supervisor | Evaluating the resilience of emerging winegrape cultivars in Australian vineyards to multiple abiotic stresses | Doctor of Philosophy | Doctorate | Full Time | Mr Caoyang Wang |
| 2025 | Co-Supervisor | Evaluating the resilience of emerging winegrape cultivars in Australian vineyards to multiple abiotic stresses | Doctor of Philosophy | Doctorate | Full Time | Mr Caoyang Wang |
| 2024 | Principal Supervisor | Characterization of hydrotropism in crop plants using barley as a model system | Doctor of Philosophy under a Jointly-awarded Degree Agreement with | Doctorate | Full Time | Ms Iza Fatima |
| 2024 | Principal Supervisor | Characterization of hydrotropism in crop plants using barley as a model system | Doctor of Philosophy | Doctorate | Full Time | Ms Iza Fatima |
| 2023 | Principal Supervisor | Finding the Missing Links in Salt and Water Transport in Plants | Doctor of Philosophy | Doctorate | Full Time | Ms Zhale Hekmati |
| 2023 | Principal Supervisor | Finding the Missing Links in Salt and Water Transport in Plants | Doctor of Philosophy | Doctorate | Full Time | Ms Zhale Hekmati |
| Date | Role | Research Topic | Program | Degree Type | Student Load | Student Name |
|---|---|---|---|---|---|---|
| 2017 - 2023 | Co-Supervisor | The Role of GABA in Plant Salinity and Hypoxia Responses | Doctor of Philosophy | Doctorate | Full Time | Miss Ying Meng |
| 2015 - 2019 | Co-Supervisor | Elucidation of the Ammonium Major Facilitator (AMF) Family in Plants | Doctor of Philosophy | Doctorate | Full Time | Mr Apriadi Situmorang |
| Date | Role | Committee | Institution | Country |
|---|---|---|---|---|
| 2017 - 2019 | Representative | Australian Society of Plant Scientists Executive | Discipline Cell Biology | Australia |
| Date | Role | Membership | Country |
|---|---|---|---|
| 2015 - ongoing | - | Society of Experimental Biology | - |
| 1998 - ongoing | Member | Australian Society of Plant Scientists | - |




