
Prof Jose Polo
Director, Adelaide Centre for Epigenetics
School of Pharmacy and Biomedical Sciences
College of Health
Eligible to supervise Masters and PhD - email supervisor to discuss availability.
Jose Maria Polo was born in Buenos Aires, Argentina where he graduated from Buenos Aires University as a Biochemist. In 2002, Jose began his graduate studies at Albert Einstein College of Medicine, New York under the supervision of Dr. Ari Melnick where he worked on the transcriptional mechanism of the BCL6 repression complex in B-cell lymphomas and B-cell maturation. In 2008, he obtained his PhD and moved to Boston to the laboratory of Dr. Konrad Hochedlinger at the Harvard Stem Cell Institute to work on reprogramming of adult cells into induced pluripotent stem (iPS) cells. In particular, his work focused on the acquisition of immortality and the existence of epigenetic memory during reprogramming. In June 2011, established his independent research group at Monash University, where he holded appointments to the departments of Anatomy and Developmental Biology and to the Australian Regenerative Medicine Institute. In 2012, Jose was awarded a NHMRC Career Development Fellowship, in 2014 a Silvia and Charles Viertel Senior Medical Research Fellowship and in 2018 a Future Fellowship to continue his work in the molecular mechanism governing the reprogramming process and the epigenetic mechanism underpinning cell fate. In October 2021, Jose was recruited to the University of Adelaide as the inaugural Director of the Adelaide Centre for Epigenetics (ACE) and group leader of the recently established South Australian Immunogenomics Cancer Institute (SAiGENCI). In Adelaide, he will continue his work in epigenetics and its application to reprogramming, early embryogenesis and cancer. His work in epigenetics, reprogramming and cancer has been published in journals such as Nature, Cell, Nature Genetics, Cell Stem Cell and Nature Medicine among others as well as recognised with several awards including the Merit Award from the American Society of Haematology, the inaugural Metcalf Award, Victorian Young Tall Poppy Award, the Monash’s Vice-Chancellor award. In 2016, he co-founded Mogrify Ltd to translate reprogramming technologies into therapies, receiving several accolades including the 2019 Scrip Innovation Award.
My Research
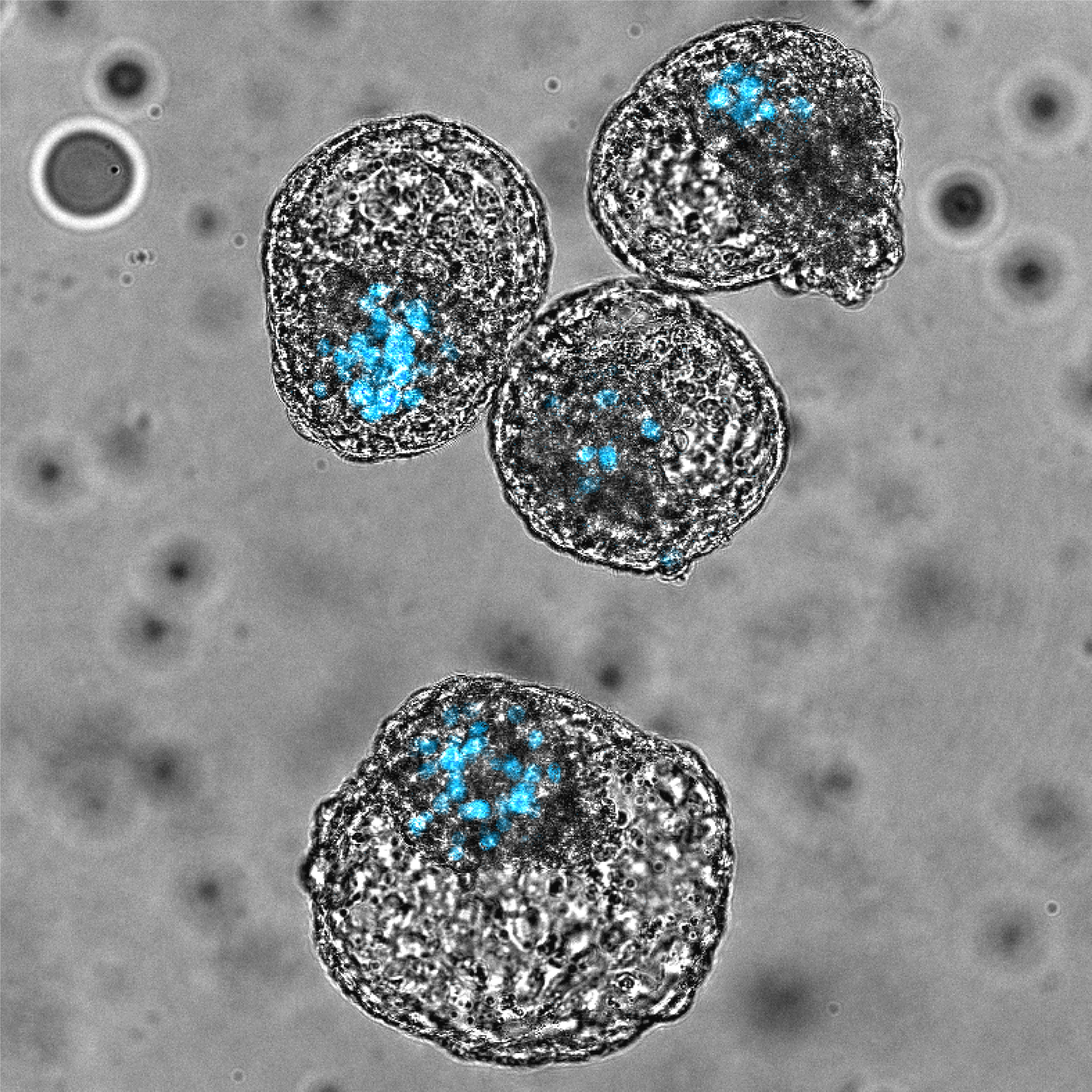
The Polo group is interested in the transcriptional and epigenetic mechanisms that govern cell identity, in particular pluripotency, reprogramming of somatic cells into induced pluripotent stem (iPS) cells, development and cancer.
Being able to reprogram any specific mature cellular program into a pluripotent state and from there back into any other particular cellular program provides a unique tool to dissect the molecular and cellular events that permit the conversion of one cell type to another. Moreover, iPS cells and the reprogramming technology are of great interest in pharmaceutical and clinical settings, since the technology can be used to generate animal and cellular models for the study of various diseases as well in the future to provide specific patient tailor made cells for their use in cellular replacement therapies. By using a broad array of approaches through the use of mouse and human stem cells models combined with different molecular, biochemical, cellular techniques and genome wide approaches, our lab will aim to dissect the nature and dynamics of such events. Understanding the events leading to the generation of iPS cells has allowed us to not only she light into this process but it has led to the generation of complex models of humans development. Furthermore, we are using and translating the tools and knowledge gained through this work into the fields of development, cancer and several other diseases.
We are particularly interested in the following aspects:
1) The kinetics and universality of the epigenetic and genomic changes occurring during cell fate transitions.
2) The composition and assembly kinetics of transcriptional regulation complexes at pluripotency genes and cancer associated transcription factors.
3) How the cell of origin influences the in vitro and in vivo plasticity potential of cells generated during the reprogramming process.
4) The role of transcription factors in cancer initiation and progression
5) Understanding mammalian development through the use of complex in vitro models
6) Synthetic cell biology

Higher Degree Research Projects
Understanding the epigenetic and transcriptional changes during reprogramming of cells to pluripotent stem cells.
The derivation of human embryonic stem cells (hESCs) and, more remarkably, the generation of human induced pluripotent stem cells (iPSCs) has revolutionised our understanding of pluripotency and opened new avenues for disease modelling, drug screening and regenerative medicine. The ability to reprogram any mature cell back to a pluripotent state, and then into another cell type, provides a unique opportunity to dissect the molecular and cellular events that occur during this conversion. Our lab aims to understand the kinetics and universality of the epigenetic and genomic changes occurring during reprogramming, the composition and assembly kinetics of transcriptional regulation complexes of pluripotency genes and how the cell of origin influences the in vitro and in vivo plasticity potential of cells generated during the reprogramming process. We will achieve this by combining stem cell technologies with several different molecular, biochemical, cellular techniques and genome-wide approaches; including ChIP, CUT&RUN, and single cell “omics”.
Molecular investigation of induced trophoblast cells (iTSC) and their derivatives.
The derivation of human embryonic stem cells (hESCs) and, more remarkably, the generation of human induced pluripotent stem cells (iPSCs) has revolutionised our understanding of pluripotency and opened new avenues for disease modelling, drug screening and regenerative medicine. However, the trophectoderm (TE) gives rise to the placenta and as such, iPSCs cannot provide models for TE or placenta. To address this important challenge, our lab has generated iTSCs for the first time. Our lab will use this model with both human and non-human primate cells, to understand the transcriptional and epigenomic changes that occur in vitro in the early human trophoblast and demonstrate that iTSCs can be used for disease modelling.
Pluripotency factors and cancer
Pluripotent stem cells can self-renew indefinitely and give rise to all cells of the adult organism. These remarkable capacities are the result of a core transcriptional network controlled by OCT4, SOX2 and NANOG, whose expression is lost upon differentiation. Several cancers have been shown to reactivate OCT4, SOX2 and/or NANOG, with high expression levels positively correlating with cancer progression and severity. Since they are linked to proliferative and multi-lineage differentiation capacity of cancer cells, they present attractive anticancer targets. However, they are considered “undruggable” since they lack catalytic active sites for molecules to bind.
To provide therapeutic alternatives, the Polo lab has adapted and developed various novel techniques to determine how expression and function of the pluripotency factors OCT4, SOX2 and NANOG are controlled in various physiological and pathological cell types, including embryonic stem cells and cancer respectively.
Uncovering the regulatory complex of Bcl6 in lymphomas
Cellular identity is controlled by transcription factors, which bind to specific regulatory elements (REs) within the genome to regulate gene expression and cell fate changes. Recent advances in epigenome profiling techniques have significantly increased our understanding of which REs are utilised in which cell type, however, which factors interact with these REs remains largely elusive. A major impediment to dissecting protein complexes at specific genomic loci is the shortage of appropriate techniques. The most common technique to assess TF binding is chromatin immunoprecipitation (ChIP), which relies on antibodies to interrogate the binding sites of a single TF. Yet, ChIP does not allow dissection of the composition of a multi-protein complex at a specific locus. Importantly, the Polo lab has developed a novel epigenetic technique termed TINC (TALE-mediated Isolation of Native Chromatin), which allows us to do exactly that (Knaupp et al., Stem Cell Reports 2020). TINC relies on epitope-tagged TALEs, which are DNA-binding proteins engineerable to target specific genomic regions. Upon cross-linking of the cells, the target regions are isolated based on affinity purification of the TALE and associated nucleic acid and protein molecules are analysed by next generation sequencing and mass spectrometry, respectively. In our proof-of-concept experiments, we dissected the protein complex formed at the Nanog promoter, a key pluripotency RE. We identified TFs previously known to bind to this locus as well as novel proteins whose role in pluripotency we further validated (Knaupp et al., Stem Cell Reports 2020). Consequently, with this valuable technique at hands, this PhD project aims at deciphering how the BTB/POZ transcriptional repressor and oncogene BCL6 is (mis)regulated in B-cell lymphomas, which in turn has major potential in identifying novel therapeutic targets.
| Date | Position | Institution name |
|---|---|---|
| 2021 - ongoing | Professor of Epigenetics and Director of the Adelaide Centre for Epigenetics | University of Adelaide |
| 2018 - 2021 | Unit coordinator for Master of Biotechnology | Australian Regenerative Medicine Institute |
| 2011 - ongoing | Professor and Group Leader in the Faculty of Medicine, Nursing and Health Sciences | Monash University |
| 2011 - 2021 | Dual appointment with the Australian Regenerative Medicine Institute (ARMI) and the Department of Anatomy and Developmental Biology (ADB) | Monash University |
| 2008 - 2011 | Research Fellow | Harvard Stem Cell Institute and Centre for Regenerative Medicine, |
| 2002 - 2008 | Graduate Student | Albert Einstein College of Medicine |
| 2000 - 2002 | Junior Lecturer | University of Buenos Aires |
| 1998 - ongoing | Visiting Student | Memorial Sloan Kettering Cancer Center |
| 1995 - 2002 | Research Assistant | University of Buenos Aires |
| 1995 - 2000 | Teaching Assistant | University of Buenos Aires |
| Date | Type | Title | Institution Name | Country | Amount |
|---|---|---|---|---|---|
| 2019 | Award | MSD’s Innovation Award at the 15th Annual Scrip Awards | Mogrify Ltd | United Kingdom | - |
| 2018 | Nomination | Selected as Next Generation of Leaders of the International Society for Stem Cell Research | International Society for Stem Cell Research | Australia | - |
| 2018 | Fellowship | ARC Future Fellowship | ARC | Australia | - |
| 2017 | Award | Vice-Chancellor’s Diversity and Inclusion Award | Monash University | Australia | - |
| 2015 | Achievement | Specialist Plenary Speaker, Australian Academy of Science’s Theo Murphy High-Flyer Think Tank, Australia | Australian Academy of Science’s Theo Murphy High-Flyer Think Tank | Australia | - |
| 2014 | Award | Metcalf Award | National Stem Cell Foundation of Australia | Australia | - |
| 2014 | Achievement | Invited as one of eight promising early career scientists (from all scientific disciplines across the world) of the Future Leaders program of the Science and Technology in Society Forum in Kyoto | Future Leaders program of the Science and Technology in Society Forum in Kyoto | Australia | - |
| 2014 | Award | Deans Award for Excellence in Research (Early Career) | Monash University | Australia | - |
| 2014 | Fellowship | Sylvia and Charles Viertel Senior Medical Research Fellowship | Sylvia and Charles Viertel | Australia | - |
| 2013 | Award | Victorian Young Tall Poppy Science Award | Victorian Young Tall Poppy Science | Australia | - |
| 2012 | Fellowship | Career Development Fellowship - NHRMC, Australia | NHMRC | Australia | - |
| 2011 | Fellowship | Larkins Fellowship | Monash Universit | Australia | - |
| 2009 | Award | Postdoctoral Fellowship Award | Massachusetts General Hospital | United States | - |
| 2005 | Award | Pre-doctoral Fellowship | National Cancer Centre | Australia | - |
| 2004 | Award | Merit Award and Plenary Speaker from the American Society of Haematology | American Society of Haematology | United States | - |
| Date | Institution name | Country | Title |
|---|---|---|---|
| 2002 - 2008 | Albert Einstein College of Medicine | United States | Doctor of Philosophy (PhD) |
| 2002 - 2004 | Albert Einstein College of Medicine | United States | Master of Science (MSci) |
| 1993 - 2000 | University of Buenos Aires | Argentina | Biochemist (Professional Degree) |
| Year | Citation |
|---|---|
| 2023 | Tan, J. P., Liu, X., & Polo, J. M. (2023). In vitro models of human blastocysts and early embryogenesis. In P. C. K. Leung, & J. Qiao (Eds.), Human Reproductive and Prenatal Genetics (2 ed., pp. 311-328). Elsevier. DOI |
| 2022 | Onfray, C., Tan, J. P., Kilens, S., Liu, X., Polo, J., & David, L. (2022). Induction of Human Naïve Pluripotent Stem Cells from Somatic Cells. In P. Rugg-Gunn (Ed.), Methods in Molecular Biology (Vol. 2416, pp. 39-51). Springer US. DOI Scopus2 Europe PMC2 |
| 2022 | Knaupp, A. S., Schittenhelm, R. B., & Polo, J. M. (2022). Characterization of Mammalian Regulatory Complexes at Single-Locus Resolution Using TINC. In J. Horsfield, & J. Marsman (Eds.), Chromatin: Methods and Protocols (Vol. 2458, pp. 175-193). New York, NY: Humana. DOI Europe PMC1 |
| 2019 | Ouyang, J. F., Kamaraj, U. S., Polo, J. M., Gough, J., & Rackham, O. J. L. (2019). Molecular Interaction Networks to Select Factors for Cell Conversion (vol 1975, 2019). In P. Cahan (Ed.), COMPUTATIONAL STEM CELL BIOLOGY: METHODS AND PROTOCOLS (Vol. 1975, pp. C1). HUMANA PRESS INC. DOI |
| 2019 | Ouyang, J. F., Kamaraj, U. S., Polo, J. M., Gough, J., & Rackham, O. J. L. (2019). Molecular interaction networks to select factors for cell conversion. In Methods in Molecular Biology (Vol. 1975, pp. 333-361). Springer New York. DOI Scopus3 WoS4 Europe PMC3 |
| 2019 | Liu, X., Chen, J., Firas, J., Paynter, J. M., Nefzger, C. M., & Polo, J. M. (2019). Generation of mouse-induced pluripotent stem cells by lentiviral transduction. In Methods in Molecular Biology (Vol. 1940, pp. 63-76). Springer New York. DOI Scopus3 WoS3 Europe PMC3 |
| 2015 | Nefzger, C. M., Alaei, S., & Polo, J. M. (2015). Isolation of reprogramming intermediates during generation of induced pluripotent stem cells from mouse embryonic fibroblasts. In Methods in Molecular Biology (Vol. 1330, pp. 205-218). Springer New York. DOI Scopus3 Europe PMC3 |
| Year | Citation |
|---|---|
| 2025 | Arthurs, A. L., Bhattacharjee, R., Smith, M. D., Medina, D., Menkhorst, E., Mora, G., . . . Roberts, C. T. (2025). Late-Onset Preeclampsia is characterised by Accelerated Placental Aging.. DOI |
| 2024 | Buckberry, S., Liu, X., Poppe, D., Tan, J. P., Faulkner, G., Polo, J., & Lister, R. (2024). Transient Naive Treatment (TNT) iPS cells do not feature Sendai virus expression: Response to Sendai virus persistence questions the transient naive reprogramming method for iPSC generation. DOI |
| 2023 | Ly, H., Chung, J., Nguyen, J. H. V., Tian, L., Schroeder, J., Knaupp, A., . . . Ryall, J. (2023). Metabolism regulates muscle stem cell self-renewal by connecting the microenvironment and histone acetylation. DOI Europe PMC1 |
| 2023 | Yap, K., Schröder, J., Gerrand, Y. -W., Kong, A., Fox, A., Knowles, B., . . . Mitchell, G. (2023). Liver-specification of human iPSC-derived endothelial cells transplanted into mouse liver. DOI |
| 2023 | Chowdhury, M. M., Zimmerman, S., Leeson, H., Nefzger, C. M., Mar, J. C., Laslett, A., . . . Cooper-White, J. J. (2023). Substrate stiffness facilitates improved induced pluripotent stem cell production through modulation of both early and late phases of cell reprogramming. DOI |
| 2023 | Healy, E., Zhang, Q., Gail, E., Agius, S., Sun, G., Bullen, M., . . . Davidovich, C. (2023). The apparent loss of PRC2 chromatin occupancy as an artefact of RNA depletion. DOI Europe PMC2 |
| 2022 | Namipashaki, A., Pugsley, K., Liu, X., Abrehart, K., Lim, S. M., Sun, G., . . . Hawi, Z. (2022). Integration of Xeno-Free Single-cell Cloning in CRISPR-mediated DNA Editing of Human iPSCs Improves Homogeneity and Methodological Efficiency of Cellular Disease Modelling. DOI |
| 2022 | Chang, Y. -C., Lisa Wong, S. F., Schroeder, J., Hauswirth, G., Shylo, N., Moore, E., . . . McGlinn, E. (2022). <i>Nr6a1</i> controls axially-restricted body elongation, segmentation, patterning and lineage allocation. DOI |
| 2022 | Gerdes, P., Lim, S. M., Ewing, A., Larcombe, M., Chan, D., Sanchez-Luque, F., . . . Faulkner, G. (2022). Retrotransposon instability dominates the acquired mutation landscape of mouse induced pluripotent stem cells. DOI |
| 2022 | Rosenbluh, J., Tuano, N., Beesley, J., Manning, M., Shi, W., Malaver-Ortega, L., . . . Chenevix-Trench, G. (2022). CRISPR screens identify gene targets and drug repositioning opportunities at breast cancer risk loci. DOI |
| 2021 | Chen, J., Neil, J. A., Tan, J. P., Rudraraju, R., Mohenska, M., Sun, Y. B. Y., . . . Polo, J. M. (2021). An iTSC-derived placental model of SARS-CoV-2 infection reveals ACE2-dependent susceptibility in syncytiotrophoblasts. DOI Europe PMC2 |
| 2021 | Tuano, N., Beesley, J., Manning, M., Shi, W., Malaver-Ortega, L., Paynter, J., . . . Rosenbluh, J. (2021). CRISPR screens identify gene targets and drug repositioning opportunities at breast cancer risk loci. DOI |
| 2020 | Huyghe, A., Furlan, G., Schroeder, J., Stüder, J., Mugnier, F., De Matteo, L., . . . Lavial, F. (2020). The comprehensive roadmaps of reprogramming and transformation unveiled antagonistic roles for bHLH transcription factors in the control of cellular plasticity. DOI |
| 2020 | Covello, G., Rossello, F., Filosi, M., Gajardo, F., Duchemin, A. -L., Tremonti, B., . . . Poggi, L. (2020). Transcriptome analysis of the zebrafish<i>atoh7−/−</i>mutant,<i>lakritz</i>, highlights Atoh7-dependent genetic networks with potential implications for human eye diseases. DOI |
| 2020 | Knaupp, A. S., Mohenska, M., Larcombe, M. R., Ford, E., Lim, S. M., Wong, K., . . . Polo, J. M. (2020). TINC - a method to dissect transcriptional complexes at single locus resolution - reveals novel<i>Nanog</i>regulators in mouse embryonic stem cells. DOI |
| 2019 | Mohenska, M., Tan, N., Tokolyi, A., Furtado, M., Costa, M., Perry, A., . . . Polo, J. (2019). 3D-Cardiomics: A spatial transcriptional atlas of the mammalian heart. DOI Europe PMC1 |
| 2019 | Keniry, A., Jansz, N., Gearing, L., Wanigasuriya, I., Chen, J., Nefzger, C., . . . Blewitt, M. (2019). Xmas ESC: A new female embryonic stem cell system that reveals the BAF complex as a key regulator of the establishment of X chromosome inactivation. DOI |
| 2019 | Grubman, A., Choo, X. Y., Chew, G., Ouyang, J., Sun, G., Croft, N., . . . Polo, J. (2019). Mouse and human microglial phenotypes in Alzheimer’s disease are controlled by amyloid plaque phagocytosis through Hif1α. DOI Europe PMC1 |
| 2019 | Grubman, A., Chew, G., Ouyang, J., Sun, G., Choo, X. Y., McLean, C., . . . Polo, J. (2019). A single cell brain atlas in human Alzheimer’s disease. DOI Europe PMC2 |
| 2018 | Tong, J., Lee, K. M., Liu, X., Nefzger, C., Vijayakumar, P., Hawi, Z., . . . Bellgrove, M. (2018). Generation of four iPSC lines from peripheral blood mononuclear cells (PBMCs) of an Attention Deficit Hyperactivity Disorder (ADHD) individual and a healthy sibling in an Australia-Caucasian family. DOI |
| 2018 | Pflueger, C., Tan, D., Swain, T., Nguyen, T., Pflueger, J., Nefzger, C., . . . Lister, R. (2018). A modular dCas9-SunTag DNMT3A epigenome editing system overcomes pervasive off-target activity of direct fusion dCas9-DNMT3A constructs. DOI |
Research Grants
| Date | Details | Title |
|---|---|---|
| 2021–2023 | NHMRC, Ideas Grant, CIA | Developing an in vitro model of a human blastocyst |
| 2021–2023 | NHMRC, Ideas Grant, CIA | Reprogramming human fibroblasts into induced trophoblast stem cells |
| 2021-2023 | ARC, Discovery Project, CIA | How do transcription factors control cell fate transitions? |
| 2020–2021 | MRFF-Coronavirus Research Response–Rapid Screening of Approved Drugs in Stem Cell Models for COVID-19 Treatment, CI | Stem cell-derived human tissue models for the identification of drugs to treat COVID-19. |
| 2020–2023 | Department of Health and Human Services Victoria, DHHS | Evaluating direct and indirect effects of SARS-CoV-2 on multiple organ systems using stem cell-derived human tissues |
| 2020-2022 | Cancer Council Victoria, PCI | Targeted reprogramming of Prostate Cancer |
| 2019–2020 | The CASS Foundation, CI | Targeting prostate cancer with maths. |
| 2018–2022 | ARC Future Fellowship |
Unveiling the epigenome dynamics through the pluripotency continuum The Fellowship supports Professor Polo salary. |
| 2018–2021 | NHMRC, CIB | Exploring and Targeting the Anti-Inflammatory Signaling Mechanisms of IL-3. |
| 2018–2020 | ARC Discovery Project, CIB | Regulatory architecture of the trunk-to-tail transition. |
| 2017-2020 | NHMRC, CIB | Leveraging genomics strategies to generate adult neurons from iPSCs and somatic cells. |
| 2017–2018 | Prostate Cancer Foundation of Australia, CI | A predictive computational framework for targeted reprogramming of castrate resistant prostate cancer. |
| 2016–2019 | NHMRC, CIA | Unveiling the human reprogramming pathway. |
| 2016–2019 | NHMRC, CIA | Using direct reprogramming to generate and rejuvenate Haematopoietic Stem Cells |
| 2015-2019 | NHMRC, CIB | Bone Marrow Endothelial Stem Cells have the capacity to form both the endothelial and haematopoietic hierarchies. |
| 2015-2017 | NHMRC, CIA | Inducing and controlling cellular plasticity. |
| 2014-2016 | NHMRC, CIA | Epigenetic and functional decline of intestinal stem cells during aging. |
| 2014–2018 | Sylvia & Charles Viertel Foundation Senior Medical Research Fellowship | The Fellowship supported Professor Polo salary from 2014-1018 |
| 2014-2016 | NHMRC, CIB | Advancing regenerative medicine by epigenetic engineering of human induced pluripotent stem cells. |
| 2014-2018 | Stem Cells Australia, Principal Investigator | |
| 2014 | ARC LIEF, Principal Investigator | This Grant underpinned the establishment of a Single Cell Genomic Centre |
| 2013-2015 | NHMRC, CIA | Determining how the germ layer of origin of adult somatic cells influences the differentiation potential of induced pluripotent stem cells. |
| 2013-2015 | NHMRC, CIA | Does nuclear reprogramming of granulocytes induce reversal of the haematopoiesis pathway. |
| 2012-2015 | NHMRC, Chief Investigator, CDF | The Fellowship supported Professor Polo salary from 2012-2013, it was discontinued upon being granted the Sylvia & Charles Viertel Foundation Senior Medical Research Fellowship. |
Teaching, Mentoring and Research Supervision
| Postdoctoral Fellows (Present) |
|---|
| Dr. Sandii Constable (Lab Manager) |
| Dr. Naiara Bediaga |
| Dr. German Moran |
|
|
| Postdoctoral Fellows (Past) |
|---|
| Dr. Christian Nefzger (Now group leader at UQ) |
| Dr. Fernando Rossello (Now Level C at University of Melbourne) |
| Dr. Kathryn Davidson (Now Project Manager at WEHI) |
| Dr. Alexandra Grubman (Now Medical Scientific Liaison at Biogen) |
| Dr. Jan Manent (Now postdoc at ARMI) |
| Higher Degree Research Graduate students |
|---|
| Monika Mohenska (Faculty Medicine, Nursing and Health Sciences PhD Program): Developing an algorithm to predict cellular identities given a spatial niche. |
| Joseph Chen (Industrial Partnership Fellowship) Using TFs to enhance induced pluripotency and direct reprogramming. |
| Jacob Michael Paynter (Faculty Medicine, Nursing and Health Sciences PhD Program): Generating human haematopoietic stem cells via transcription factor mediated reprogramming. |
| Michael Larcombe (Faculty Medicine, Nursing and Health Sciences PhD Program): Dissecting the regulatory complexes controlling pluripotency factors. |
| Jia Ping Tan (Faculty Medicine, Nursing and Health Sciences PhD Program): Understanding the molecular control and cell fate transitions of human induced trophoblast stem cells. |
| Esther Louise Miriklis (Australian Regenerative Medicine Institute): Spatial gene regulation via single cell analyses and advanced fluorescence microscopy. |
| Postgraduate students trained to date (PhD) | Year |
|---|---|
| Xiaodong Ethan Liu (Monash Postgraduate Award Scholarship): Integrative molecular analyses reveal distinct reprogramming trajectories into states of naïve and primed human induced pluripotency. He was awarded the top 100 thesis from a Chinese student abroad by the Chinese government in 2019. Awarded the Stem Cell Biology Ridolfo award for best student of the year in 2018. Due to COVID19, he is doing a short postdoc in the Polo Lab. | 2019 |
| Xin Yi Choo (International Postgraduate Research Scholarship): Development of novel therapeutic approaches for treatment of Alzheimer’s disease. Now postdoc at Duke-NUS, Singapore. | 2018 |
| Jaber Firas: Towards an understanding of induced cellular plasticity. He was awarded the VC thesis Commendation. Awarded the Stem Cell Biology Ridolfo award for best student of the year in 2016. Now training as MD to become a Physician-Scientist. | 2018 |
| Suzan DeBoer: Modelling epigenetic dysregulation in developmental disorders. Now a postdoc in Leiden University Medical Center, Holland. | 2017 |
| Sara Alaei: Dynamics of transcription factor targeting and chromatin states during reprogramming. Now Postdoc at ARMI | 2016 |
| Bianca Borchin: Directed derivation and FACS-mediated purification of PAX3+/PAX7+ skeletal muscle precursors from human pluripotent stem cells. Professor Polo supervised her last year and thesis writing due to her first supervisor leaving the Institute. Bianca is now a Postdoc in Memorial Sloan Kettering. | 2015 |
| Honours students trained to date | Year |
|---|---|
| Kayla Wong | 2018 |
| Jia Tan | 2018 |
| Matt Tiedemann | 2017 |
| Amber Rucinski | 2016 |
| Nathalia Tan | 2015 |
| Mitchel de Souza | 2014 |
| Jaber Firas | 2013 |
| Masters Student | Year |
|---|---|
| Daniela Pufleau Valdes (Master of Biomedical & Health Science): Molecular and functional characterisation of iTSCs derived from reprogrammed human fibroblasts. | 2019 - 2020 |
| International Masters student | Year |
|---|---|
| Frida Forsberg (Uppsala University) | 2015 |
| Research in Action undergraduate student | Year |
|---|---|
| Neiloy Chowdhury | 2018 |
| Raymond Zhang | 2018 |
| Sheree Chen | 2018 |
| Pranjal Patel | 2018 |
| Natalie Saunders | 2017 |
| Kayla Wong | 2017 |
| Kesh Faye-Chauhan | 2013 |
| Laura Corfield | 2013 |
| Erica Min Joung Kim | 2012 |
| Research Assistant |
|---|
| Dr. Guizhi Sun |
| Research Assistants (Past) |
|---|
| Dr. Melissa Holmes |
| Ketan Mishra |
| Margeaux Hodgson-Garms |
| Joseph Chen |
| Jacob Paynter |
| Michael Larcombe |
| Date | Role | Research Topic | Program | Degree Type | Student Load | Student Name |
|---|---|---|---|---|---|---|
| 2024 | Co-Supervisor | The role of coding and non-coding RNA in atherosclerotic plaque phenotype in diabetic vs non-diabetic patients with peripheral arterial disease | Doctor of Philosophy | Doctorate | Part Time | Dr Benjamin Thurston |
| 2024 | Co-Supervisor | The role of coding and non-coding RNA in atherosclerotic plaque phenotype in diabetic vs non-diabetic patients with peripheral arterial disease | - | Doctorate | Part Time | Dr Benjamin Thurston |
| 2023 | Principal Supervisor | Somatic cell reprogramming of fibroblasts from different mammals to model early development. | Doctor of Philosophy | Doctorate | Full Time | Miss Elly Deidre Walters |
| 2023 | Principal Supervisor | How pluripotent transcription factors reshape the prostate cancer epigenetic landscape to reprogram the cell fate toward neuroendocrine-like with their cofactors | Doctor of Philosophy | Doctorate | Full Time | Mr Tianjun Zhang |
| 2023 | Principal Supervisor | Somatic cell reprogramming of fibroblasts from different mammals to model early development. | Doctor of Philosophy | Doctorate | Full Time | Miss Elly Deidre Walters |
| 2023 | Principal Supervisor | How pluripotent transcription factors reshape the prostate cancer epigenetic landscape to reprogram the cell fate toward neuroendocrine-like with their cofactors | - | Doctorate | Full Time | Mr Tianjun Zhang |
| 2023 | Principal Supervisor | Somatic cell reprogramming of fibroblasts from different mammals to model early development. | - | Doctorate | Full Time | Miss Elly Deidre Walters |
| Date | Role | Committee | Institution | Country |
|---|---|---|---|---|
| 2020 - ongoing | Chair | ARMI Group Leader Meeting | ARMI | Australia |
| 2019 - ongoing | Chair | Academic Steering Committee | Monash Micromon Genomics Platform | Australia |
| 2019 - ongoing | Member | Co-organiser of the Single Cell Symposium, | DUKE-NUS, Singapore | Singapore |
| 2019 - ongoing | Member | International Society for Stem Cell Research (ISSCR) Education Committee | International Society for Stem Cell Research (ISSCR) | Australia |
| 2019 - ongoing | Member | Monash Proteomics Platform Steering Committee | Monash Proteomics | Australia |
| 2017 - ongoing | Co-Chair | Reprogramming Symposium | Stem Cells Stream at ComBio - ASBMB 2017 | Australia |
| 2017 - 2019 | Member | Pluripotency Stream Leader and member of the Scientific Leadership Group at | ARC Stem Cells Australia Special Initiative. | Australia |
| 2016 - 2018 | Member | Department of Anatomy and Developmental Biology Executive Committee | Department of Anatomy and Developmental Biology | Australia |
| 2016 - ongoing | Chair | Chair and Programme Co-organizer, Margaret River Regional Forum/ASSCR, 2017 | Margaret River Regional Forum/ASSCR | Australia |
| 2015 - 2018 | Member | ARMI HDR committee | ARMI | Australia |
| 2014 - ongoing | Chair | Lead organizer of the Australasian Society for Stem Cell Research Annual Meeting. | Australasian Society for Stem Cell Research | Australia |
| 2014 - ongoing | Chair | Pluripotency Symposium, Stem Cells Stream at ComBio - ASBMB 2014 | Stem Cells Stream | Australia |
| 2014 - 2017 | Member | Faculty of Biomedical and Psychological Sciences (FBPS) Gender Equity Committee | Faculty of Biomedical and Psychological Sciences (FBPS) | Australia |
| 2013 - 2018 | Member | MHTP Bioinformatics Committee | Monash Institute of Medical Research | Australia |
| 2013 - 2017 | Member | Cell Reprogramming Australia | Cell Reprogramming Australia | Australia |
| 2013 - 2015 | Co-Chair | Cell Reprogramming Australia Annual Conference | Cell Reprogramming Australia | Australia |
| Date | Role | Editorial Board Name | Institution | Country |
|---|---|---|---|---|
| 2020 - ongoing | Member | Stem Cell Reports (Official Journal of the International Society for Stem Cell Research) | International Society for Stem Cell Research | Australia |
| Date | Title | Type | Institution | Country |
|---|---|---|---|---|
| 2016 - ongoing | Silvia and Charles Viertel Foundation (External) | Peer Review | Silvia and Charles Viertel Foundation (External) | - |
| 2016 - ongoing | Biotechnology and Biological Sciences Research Council (UK) | Peer Review | Biotechnology and Biological Sciences Research Council (UK) | - |
| 2013 - ongoing | Fondation pour la Recherche Médicale (France) | Peer Review | Fondation pour la Recherche Médicale (France) | - |
| 2013 - ongoing | Reviewer for Academic Promotion | Peer Review | University of Melbourne, University of Edinburgh, Tokyo University, MRC LMS, Imperial College, John Hopkins University | - |
| 2012 - ongoing | Neurological Foundation of New Zealand | Peer Review | Neurological Foundation of New Zealand | - |
| 2011 - ongoing | NHMRC (Panel member and external) and ARC. | Peer Review | NHMRC and ARC | - |





